Mid-Term Plan
TOP>Investor Relations>Mid-Term Plan
JCR Pharmaceuticals Co., Ltd. (TSE 4552; Chairman and President: Shin Ashida; “JCR”) announced that JCR has formulated the Midterm Business Plan for FY2023-FY2027, “Reach Beyond, Together,” and hereby provides an overview.
JCR's Corporate Philosophy is “Contributing towards people's healthcare through pharmaceutical products.” Based on this philosophy, JCR has since its foundation in 1975 advanced business activities through its original “R&D” and “Manufacturing” to deliver world-first medicines with new mechanisms to patients, including TEMCELL® and IZCARGO®.
JCR is now one of the few companies in Japan that can operate the entire process of biopharmaceuticals, from research through to manufacturing. Having built foundational “R&D” and “Manufacturing” capabilities, we will continue to deliver value only JCR can provide to patients with rare diseases and their families.
Overview of Midterm Business Plan for FY2023-FY2027
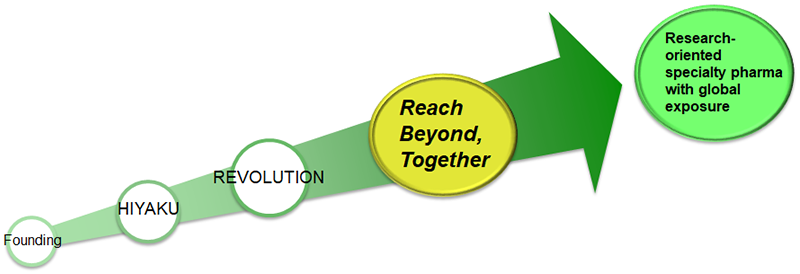
“Reach Beyond, Together”
Keyword
Reach Beyond, Together
〈Positioning of this Plan〉
Under the newly formulated Midterm Business Plan for FY2023-FY2027 “Reach Beyond, Together,” JCR will further augment the strengths we have developed through Midterm Business Plan for FY2015-FY2019 “HIYAKU” (Leap Into the Future) and Midterm Business Plan for FY2020-FY2022 “REVOLUTION” and create medicines that can only be made by JCR through its innovative drug creation platform technologies, making it a research-oriented specialty pharma with global exposure.
The keyword, “Reach Beyond, Together,” express the unity of “Team JCR” as it moves toward a common goal, as well as the platform technology J-Brain Cargo® penetrating the blood-brain barrier and making progress toward new hope together with patients and their families.
Our Credo
Having built foundational “R&D” and “Manufacturing” capabilities, we can deliver value only JCR can provide to patients with rare diseases and their families.
Five Initiatives
JCR will focus on the following five initiatives in this Midterm Business Plan.
[1] Creation of innovative core technologies
JCR will aim to create breakthrough therapies not limited to lysosomal storage disorders (LSDs) using a basic concept of J-Brain Cargo®, a platform technology JCR developed that was demonstrated as the first to penetrate the human blood-brain barrier.
[2] Demonstrating of global standard production capacity
[3] Expansion of global quality assurance system in terms of quality and quantity
“MONOZUKURI (Manufacturing)” has been a strength JCR has cultivated since foundation and it further strengthen this area to construct a resilient global supply chain capable of stable supply for any aspect or product.
[4] Early launch of products for rare diseases
Of the 17 development pipelines for LSD therapies that apply J-Brain Cargo® technology, on which R&D is moving forward, JCR aims to begin clinical trials for 5 items.
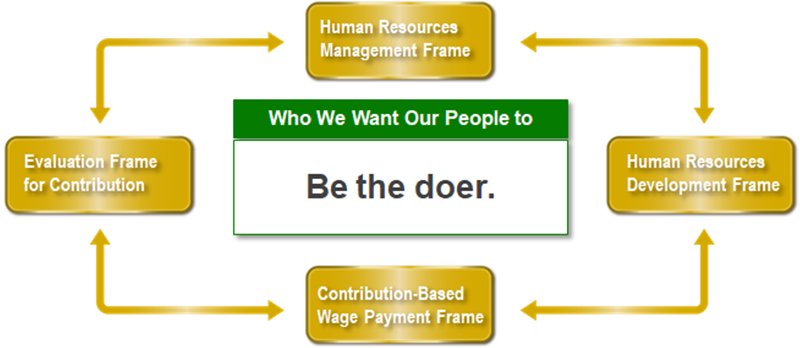
[5] Human resource development to support growth
JCR continues investment in human capital, and establish a human resource strategy that helps to improve corporate value. JCR is also building a human resource management frame as a framework to realize the image of who we want our people to become.
Sustainability
JCR will achieve of sustainability through business activities based on the areas of Rare Diseases, Environment, Society and Corporate Governance.
 Rare Diseases:
Rare Diseases:
Rare diseases, in particular, are closely related to JCR's business activities and are a domain to which JCR can especially contribute.
We will proactively engage in things that only JCR can provide even for ultra-rare diseases where there are only few patients as we aim to achieve a society where no one will be left behind.
- Development of drugs for ultra-rare diseases and construction of new business models
- Efforts to improve access to medicines (Named Patient Supply) to deliver innovative medicines to people around the world
 Environment:
Environment:
- Investigation and implementation of measures to reduce environmental impact through business activities
- Promote environmentally friendly procurement on the premise of ensuring the quality and stable supply of pharmaceuticals
 Society:
Society:
- Address the challenge of unmet medical needs by creating platform technologies
- Develop human resources so that each member of “Team JCR” can shine in their own station
 Corporate Governance:
Corporate Governance:
- Pursue governance in line with the company’s growth
- Ensure thorough compliance and strengthen risk management
* Please refer to the presentation material for details of the content.






